The clinical benefit of the new arterial tree model — mission accomplished.
*
Providing certainty about the individual risk profile for cardiovascular diseases within minutes.
The clinical benefit of the new arterial tree model
Mission accomplished
Project goal achieved: Based on a simulation model, the specific vascular properties of each patient can be read individually from the arterial tree for the first time. With unprecedented precision and thus with spectacular benefits for cardiovascular diagnostics and therapy.
This puts current standard arterial diagnostics to the test: compared with Model-based Pulse Wave Analysis (mbPWA), previous gold standards of vascular measurement literally look “old”.
A direct look into the vascular system
The ingenious thing about the measuring principle of Model-based Pulse Wave Analysis (mbPWA): Once the individual arterial model — the electronically generated ‘clone’ of the arterial tree of a real person — has been created with the help of special software, the actual physical parameters can be read off in simulatione for the first time. Several well-known parameters — including, in particular, vascular stiffness — are available within the framework of the determination via our individualized model, contrary to earlier auxiliary parameter data sets, in a quality and plausibility never before achieved. In the course of time and with an increasing number of subjects, it has become more and more apparent that the model parameter sets determined for each individual provide a whole wealth of clinically valuable information. Thus, among other things, central blood pressure can now also be read in the model in a new quality.
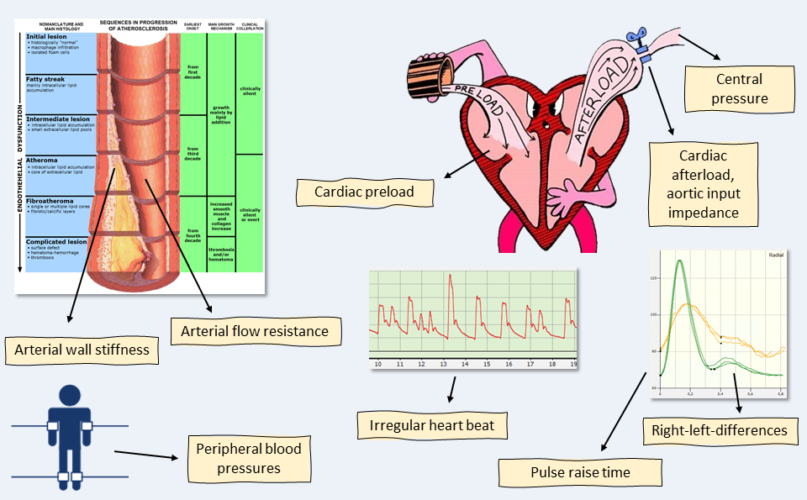
What we measure
The new parameters at a glance
- Flow resistance index of the large peripheral arteries as a measure of permeability.
- Arterial wall elasticity index
- Blood mass inertia index as a measure of blood fluidity
- Pressures directly measurable in the model, the most important being the aortic pressure (cBP)
- LVPT (Left Ventricular Plateau Time) as a measure of cardiac preload, increased, for example, in hyperhydration (especially in dialysis patients), but also in prolonged negative stress
- Aortic Input Impedance Index: the indicator of high cardiac afterload (for example, in vascular damage such as atherosclerosis)
Know More — Protect Better
The clinical benefit: New options for diagnosis and therapy
The model-based pulse wave method is a groundbreaking procedure in that the new measurement system can now actually determine arterial properties directly with the help of artificial intelligence — and yet entirely without invasive procedures that are stressful for the patient, such as an examination with a cardiac catheter. From the pre- and afterload of the heart to the elasticity of the arteries and right-left differences in peripheral blood pressure, the diagnostic options of Model-based Pulse Wave Analysis are already immense based on the large number of parameters measured — and are constantly expanding as part of ongoing studies.
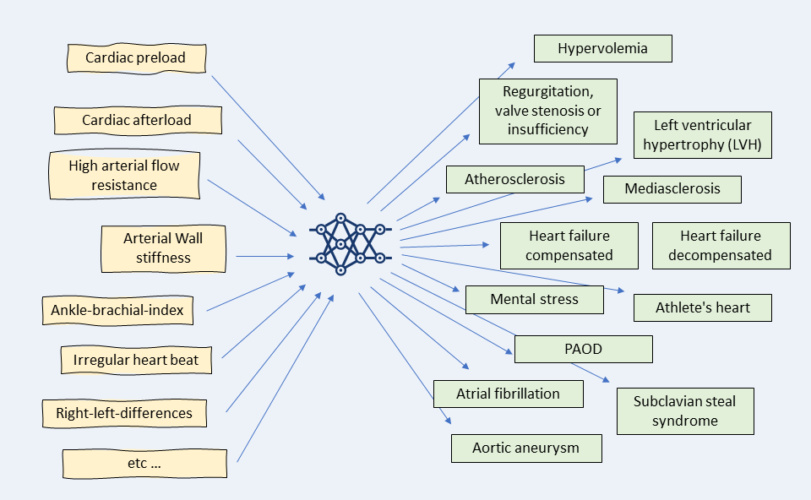
Input and Output: The diagnostic spectrum
Future application field: Preventing dialysis patients from cardiovascular damage
Since we can simulate the heart’s function within the arterial tree model, we can also determine cardiac afterload — and even preload. The afterload refers to the pressure prevailing in the aorta and represents the resistance against which the heart must pump continuously. The preload, in turn, is subject to venous return to the heart. If both — that is, the volume and pressure load on the heart — are permanently elevated, this leads to long-term damage, such as left ventricular hypertrophy (LVH). This occurs strikingly often in dialysis patients and leads to alarmingly high mortality rates in this patient group. LVH is directly related to coronary artery disease and is considered a cardiovascular high-risk factor for myocardial infarction, heart failure, or sudden cardiac death. Close monitoring with VASCASSIST, applying Model-based Pulse Wave Analysis (mbPWA), ensures that the cardiovascular system of dialysis patients, which is working under increased pressure as a result of dialysis, is protected from damage by close monitoring.
Cardiovascular parameters are often still too infrequently monitored specifically in dialysis patients.